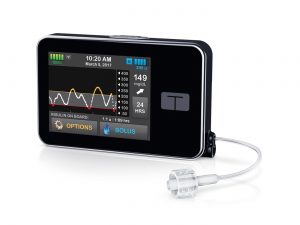
 Lots of news has been surfacing lately about the Tandem t:slim X2 pump which is being brought to Australia by AMSL. There are still some details yet to be clarified, but here’s a little recap.
Lots of news has been surfacing lately about the Tandem t:slim X2 pump which is being brought to Australia by AMSL. There are still some details yet to be clarified, but here’s a little recap.
The pump is approved by the TGA for supply in Australia. The consumables are not yet available through the NDSS IPC list (I suspect there may be an update to that at the beginning of August).
It can’t be funded through private health insurance yet. The next update to the official Prostheses List should be published by early September, and we assume the t:slim X2 will be on the new list (which should become effective in mid-September) which will enable purchases from that date on.
 However I understand some AMSL customers have been supplied with Animas Vibes as stop-gap “new” pumps until the t:slim was available. As soon as consumables are available through NDSS (see above) I suspect some of those customers will be upgraded to new t:slim pumps for the remainder of their warranty period. So we might be seeing user feedback next month!
However I understand some AMSL customers have been supplied with Animas Vibes as stop-gap “new” pumps until the t:slim was available. As soon as consumables are available through NDSS (see above) I suspect some of those customers will be upgraded to new t:slim pumps for the remainder of their warranty period. So we might be seeing user feedback next month!
CGM receiver
 The t:slim X2 is able to act as a Dexcom G5 receiver, showing you the CGM results and giving you alarms as appropriate. This feature is undoubtedly going to be available in the pump when it ships in Australia.
The t:slim X2 is able to act as a Dexcom G5 receiver, showing you the CGM results and giving you alarms as appropriate. This feature is undoubtedly going to be available in the pump when it ships in Australia.
When the Dexcom G6 is eventually approved by the TGA for supply in Australia it is expected that this will be supported by the Tandem pump.
Basal-IQ
Last month the FDA approved Tandem’s “Basal-IQ” firmware update for the pump in the US, which adds support for the Dexcom G6 as well as giving predictive-low-glucose-suspend. Otherwise known as a “hypo minimiser”.
 If you’re using the G6 CGM and you’ve enabled the Basal-IQ function, the pump extrapolates what it predicts your glucose level will be 30 minutes into the future, and if that will be 4.4 mmol/l or below, the pump suspends basal insulin delivery. Once it thinks your levels are coming up again it resumes delivery.
If you’re using the G6 CGM and you’ve enabled the Basal-IQ function, the pump extrapolates what it predicts your glucose level will be 30 minutes into the future, and if that will be 4.4 mmol/l or below, the pump suspends basal insulin delivery. Once it thinks your levels are coming up again it resumes delivery.
This function will be a massive benefit for many pump users, and hopefully will provide even smoother operation than the hypo minimiser function in Medtronic’s 640G pump.
In the US this Basal-IQ update is expected to roll out in late July / early August. To get the firmware update, there are new steps that users have to go through. Because this update involves the computer making new decisions about the insulin supply, users have to get “clinical need” paperwork signed off by their doctors and then complete official training on the new features (which can be done online). Once all that is done, each user receives an individualised access code which allows them to download the software and update their pump.
Australian firmware
We do NOT yet know what firmware features have been approved by the TGA for the initial rollout of the pump in Australia. This firmware will have new features (e.g. mmol/l units) which have not been available in the US version of the pump.
Because the Dexcom G6 is not yet approved for supply in Australia, it seems unlikely that the Basal-IQ functionality will be included in the initial release of the pump. Tandem did do trials and prepare a Basal-IQ-with-G5 submission for the FDA, but when the G6 came out they revised this to be G6-only.
We also do not know what the process of future firmware updates will be for Australian users. If the Basal-IQ functionality is not available at first, when will it get approval for supply in Australia?
Tandem have stated that firmware updates in the US will be free until the end of 2018. No-one has said anything about what will happen 6 months from now. And AMSL have not said anything about the future approval and upgrade process for Australia.
Control-IQ
The next big firmware update on the horizon seems to be Tandem’s “Control-IQ” which will be their initial closed-loop system, using the G6 CGM and Type Zero‘s loop algorithm. With this the system will not only suspend insulin delivery to avoid hypos, it will increase insulin delivery to avoid hyperglycaemia.
Trials of this are currently under way in the US, so no-one knows what the timeframe will be for this eventually getting out to users, let alone to Australia.
Apidra insulin issues
Only two insulins are labelled for use in pumps in Australia: Humalog and NovoRapid. However there are some people who are using the fast-acting Apidra in pumps off-label (usually as prescribed by their doctor). Apparently this insulin had enough issues with occlusions and blockages during Australian trials that it was only labelled for use in pens and syringes. It’s certainly known to be more temperature-sensitive than Humalog or NovoRapid. But anecdotal evidence is that it’s been working great in pumps for some Australians for years. Incidentally it is approved for use in pumps in the USA, although for much shorter intervals than the other insulins.
 Personally I’ve been using Apidra in Veo and Combo pumps for the last month, but I have been carefully replacing the pump reservoirs every 2 days to avoid any occlusion/effectiveness issues (I carry my pump in a pocket where it gets quite warm). It has been working well in my closed-loop pump system (which can take advantage of the different absorption profile) although my trial is ongoing. I’ll publish info about that exercise later.
Personally I’ve been using Apidra in Veo and Combo pumps for the last month, but I have been carefully replacing the pump reservoirs every 2 days to avoid any occlusion/effectiveness issues (I carry my pump in a pocket where it gets quite warm). It has been working well in my closed-loop pump system (which can take advantage of the different absorption profile) although my trial is ongoing. I’ll publish info about that exercise later.
Where this is relevant to the t:slim X2 is that the Tandem pump uses a radically different reservoir system, where the reservoir is a loose bladder inside the pump. Reports from US users over the past few years indicate that Apidra in this pump is especially susceptible to occlusion issues. So that would be an extra complication for anyone considering this off-label use in Australia.
Where to from here?
Hopefully answers to some of these questions will become clearer as we move closer to the actual release of this pump in Australia. It’s all quite exciting though, and I think there will be many happy pump users later in 2018.
I do also expect there to be more news about other pump options at the same time…
