As you will no doubt have heard, the choices for insulin pumps in Australia have been changing lately. There are some things we know, some we’re pretty sure of, and some we’re guessing at. This is my attempt at a summary for early February 2018.
This article has been superseded by our August 2018 pump update!
Pumps currently on the market
Medtronic 640G
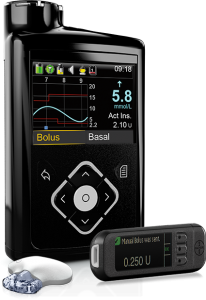 When paired with Medtronic’s Enlite2 CGM, the 640G has what they call SmartGuard, which is Predictive Low Glucose Suspend (PLGS). It suspends basal insulin delivery when the CGM indicates an approaching hypo, and this has been very useful for many people.
When paired with Medtronic’s Enlite2 CGM, the 640G has what they call SmartGuard, which is Predictive Low Glucose Suspend (PLGS). It suspends basal insulin delivery when the CGM indicates an approaching hypo, and this has been very useful for many people.
Accu-Chek Combo System
 The system is comprised of the Spirit Combo pump and the Bluetooth-connected Performa Combo meter, which acts as a remote control (so the pump can stay tucked away under clothes for example).
The system is comprised of the Spirit Combo pump and the Bluetooth-connected Performa Combo meter, which acts as a remote control (so the pump can stay tucked away under clothes for example).
Cellnovo
 This little pump attaches to your body right next to the cannula, connected by Bluetooth to a phone-size controller. It was introduced into Australia in November 2017, and there’s not a large user base yet. There has been talk of adding support for a Dexcom CGM into the controller, but no sign of that update yet.
This little pump attaches to your body right next to the cannula, connected by Bluetooth to a phone-size controller. It was introduced into Australia in November 2017, and there’s not a large user base yet. There has been talk of adding support for a Dexcom CGM into the controller, but no sign of that update yet.
Animas Vibe
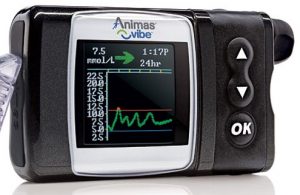 Well, it’s sort-of still on the market. After Animas announced it was closing down worldwide operations, the local agent (AMSL) is continuing support of their current Animas customer base, but has indicated they will be introducing a new pump. My understanding is that new people signing up would be started on a Vibe and transitioned to the new pump when it became available. But what that new pump would be has been a bit of a mystery. Keep reading….
Well, it’s sort-of still on the market. After Animas announced it was closing down worldwide operations, the local agent (AMSL) is continuing support of their current Animas customer base, but has indicated they will be introducing a new pump. My understanding is that new people signing up would be started on a Vibe and transitioned to the new pump when it became available. But what that new pump would be has been a bit of a mystery. Keep reading….
Internet forums have been full of questions for the last month asking things like “I’m due for a new pump later this year. What should I get?” And people bemoaning that Medtronic was the only player left (they’re obviously not!). The sensible answer is obviously “ask again when it’s closer to your rollover date and we’ll see what’s available then”. But people are always curious!
Pumps we may see soon
So what other pumps should we expect? Through a bit of sleuth work it’s not hard to make some predictions. So here goes.
Ypsopump

The Ypsopump (from Ypsomed) has TGA approval for sale in Australia, and I gather the plan includes rolling it out to initial patients in the next few months.
But what about AMSL’s new pump? There have been a few wild guesses doing the rounds of the internet, but some new information has just come to light this week!
Tandem?
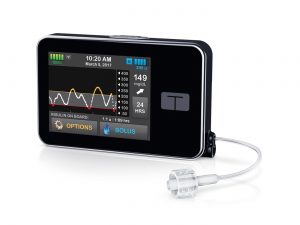 One strong contender in the rumour mill has been the Tandem t:slim X2 pump, which already integrates with Dexcom’s G5 CGM (note that AMSL are the Australian Dexcom agent). Recently Tandem announced they were expanding to markets outside the US, and the timing seemed suspicious. However the Tandem pump does not yet have TGA approval for sale in Australia, so if the Tandem is coming then presumably the application process would be underway already. Time will still tell if anything comes of that.
One strong contender in the rumour mill has been the Tandem t:slim X2 pump, which already integrates with Dexcom’s G5 CGM (note that AMSL are the Australian Dexcom agent). Recently Tandem announced they were expanding to markets outside the US, and the timing seemed suspicious. However the Tandem pump does not yet have TGA approval for sale in Australia, so if the Tandem is coming then presumably the application process would be underway already. Time will still tell if anything comes of that.
Incidentally the X2 pump can have software updates applied by the user, and updates expected in 2018 include support for Dexcom’s G6 CGM, and Tandem’s Basal-IQ PLGS system. They have run some trials in the US using the X2 in a hybrid closed-loop mode (again using the G6), but there’s no indication of when such a feature might finally arrive on the market.
OmniPod?
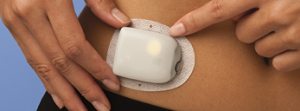 Another rumour has guessed that AMSL might introduce the OmniPod patch pump. However there are a couple of issues with this rumour:
Another rumour has guessed that AMSL might introduce the OmniPod patch pump. However there are a couple of issues with this rumour:
- The OmniPod received TGA approval way back in 2010, but the company that represents them in Australia has not managed to bring it to market in the last 8 years.
- The OmniPod doesn’t fit the “prosthetic pancreas” funding model used in Australia. For most pumps the major cost is in the pump device itself, which is covered by private health insurance in a hospital visit. The ongoing cost of consumables (reservoirs and cannulae) is not covered by insurance, but is subsidised by the government through the NDSS. But the OmniPod would turn this model on its head. A PDM (“Personal Diabetes Manager”) controller is supplied when you start, but then instead of inserting a new cannula every 3 days, you use a whole new pump every 3 days. I’ve said for a long time that I don’t think the NDSS funding model can support this. Not only does the ongoing cost of pumps outweigh the cost of cannulas, I think the total cost of the system is actually more than the traditional “tubed” pump systems.
Medtrum A6 TouchCare
 A new pump you might not have heard of! Check it out.
A new pump you might not have heard of! Check it out.
Note: an editorial mistake initially had “CareSense” in the name. My apologies!
Like the OmniPod this is a “patch pump”, where the pump is attached to the skin without any intervening tubing, and a “PDM” controller has the buttons and display. It also has its own CGM, apparently with a PLGS implementation in the pump. But unlike the OmniPod, the pump that is worn is made up of two parts, one of which is replaced regularly (it has the insulin reservoir, the cannula, and the battery) while the logic and control side of the pump is re-used. This might be something that can be shoe-horned into the Australian funding model.
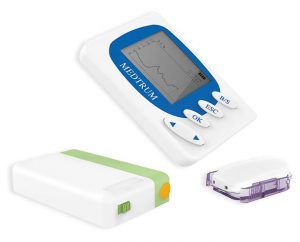 Just last week new TGA approvals were issued for two devices: the A6 TouchCare, and the reservoir. Now as we’ve seen with the OmniPod, getting a device approved by the TGA is just the start, and doesn’t guarantee the device will actually reach the market. But it’s a start!
Just last week new TGA approvals were issued for two devices: the A6 TouchCare, and the reservoir. Now as we’ve seen with the OmniPod, getting a device approved by the TGA is just the start, and doesn’t guarantee the device will actually reach the market. But it’s a start!
Interestingly, the company name attached to those approvals: Emergo. This name is probably familiar to everyone who receives Dexcom CGM supplies from AMSL: it’s printed on stickers on those boxes. Does this mean that the Medtrum A6 is the new pump that will be introduced by AMSL in 2018? It certainly seems possible! Of course Emergo have also been sponsors of 918 other TGA approvals, so it’s possible they’re doing it for another company. But that seems unlikely in the domain of insulin pumps.
There have also been rumours of two AMSL pump options, so who knows what else might appear in the coming months?
While the A6 TouchCare has its own S6 CGM (which apparently buffers data unlike the Dexcom G4/G5) it’s unclear whether that was part of the TGA approval issued for the system. We’ll have to wait and see whether AMSL integrates the A6 with their Dexcom offering somehow.
Pumps in limbo
There are yet more pumps in the offing, but we don’t know if/when we’ll see them:
DANA RS
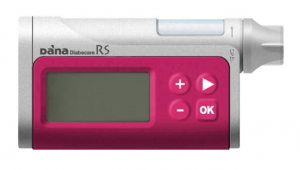 This pump is very interesting to many people, but is apparently currently waiting to get onto the Australia “prosthesis list” and thus be able to be funded through private health insurance like the other pumps. The RS is an update to the earlier DANA R pump, and while both have Bluetooth which allow Android phones to connect to them, the RS can also be connected to Apple devices. It will be interesting to see if the DANA RS makes it onto the official list, and what support infrastructure DANA’s Australian agent is able to put into place. No-one has any date estimates. At least not until the prosthesis list gets updated.
This pump is very interesting to many people, but is apparently currently waiting to get onto the Australia “prosthesis list” and thus be able to be funded through private health insurance like the other pumps. The RS is an update to the earlier DANA R pump, and while both have Bluetooth which allow Android phones to connect to them, the RS can also be connected to Apple devices. It will be interesting to see if the DANA RS makes it onto the official list, and what support infrastructure DANA’s Australian agent is able to put into place. No-one has any date estimates. At least not until the prosthesis list gets updated.
Medtronic 670G
This is Medtronic’s first hybrid closed-loop pump system, but while it’s available in the US, it’s still involved in trials here in Australia, and I don’t expect to see it arrive here in 2018.
There you go, that’s about all we know at the moment! Stay tuned as the market keeps developing during the year!

Thanks. Great information. Looking for info for my 15y.o son, not pumping yet. Previously was adamant not to have a pump but since diabetic camp he is thinking about. Now keen for a libre but might wait until on the ndss.
Thanks David, a good summary, hope things move faster than anticipated!
I know I love my Medtronic 670G. It is great to see others manufacturers entering the international market.
I want one! Why is Australia sooo sloooow!
Great summary David! Thanks
Excellent Overview.
I’m on the Vibe and didn’t realise till recently I’m 9 months out of warranty. I need something soon before I have any issues.
Cellnovo is my choice for what is currently on the market but I’ll hold out a little longer to see what AMSL do. The reps are very tight lipped and they most likely don’t even know. I’m hoping it’s the T:slim although the Touchcare looks interesting. I think anything will be an improvement on the Vibe.
Its going to be the t slim.
Thanks for keeping us up to date David
Wow, exciting times ahead. I now understand the fundamental difference with Cell novo (replaceable pump & all). Before I only knew it was tubeless. A smaller pump such as cell novo will be interesting to watch this space…Very informative, thank you.
Apologies, I meant differences for Omnipod! Tired D mamma…
Be careful with the Dana RS pump. If it is anything like the Dana R it is not worth the cost or the usage. Mine is sitting in its box and I am back to my Vibe. The supplier of it shut his website down and left the country. Many other users have been left amazed by this. The pump uses its own batteries which can only be purchased by buying an infusion set. It broke and when he eventually sent it back to Korea it took 3 months to have it repaired and returned.
Indeed if the DANA RS gets onto the Prostheses List, they still have the hurdle of getting people (including DEs) willing to promote it as a solution. They got themselves into a mess with the R, and will have to build up trust again for whatever supplier takes it on.
None the less, the Dana pump is blue tooth enabled and connects to smartphone. How long before the libre connects to it? Larger companies say the Dana is hackable. I say so what!
In the months since this article, the DANA RS has in fact got onto the Prostheses List and is being bought by people through health insurance.
The pump isn’t “hackable” as such, but it is able to communicate with AndroidAPS.
It’s one of two pumps available new that can be used in a loop.
I have a Medtronic 640G. I’m not impressed by its size and weight. It’s my first pump and after a few years of wearing one I’m still not loving it. I’d be looking at anything smaller when it comes time to switch.
Thanks for the review. It helped me a lot. Im just thinking to start pumping.. but none is available in my country. They used to have one, but it seems that they stopped it due to thw very high cost
I was looking forward to getting the 670g and the “deal” where they offer the new sensors for 6 months at half price, that is, $125/month. But, it seems like I will fall between the cracks in the system. The pump warranty for my current Medtronic 640g runs out on the 20th April. This deal finishes on the 26th April. I cannot apply for the new pump until the warranty runs out. My health fund takes two weeks to review the request and issue the “buy” order to Medtronic who then communicate with the Diabetes Educator to schedule a fitting and education date. Result: I will have to continue to pay $250/month for sensors. This is expensive when one is on a pension. I was looking forward to saving $750.
Great article.
I am so keen to get a tubeless insulin loop like that of the A6. I work with trees an bush regeneration so having a tube between devices is just not a safe option. I to have been using the Dexcom 5 device with a promise that something new was coming to the market in late 2018 and due to regulation laws this never happened in Australia. I am desperate for new technology to become available in Australia.
Please continue with your articles. It helps to get an overall insight without any business ideologie comforts.
Many thanks
Todd Stark
hey is the cell novo on the prostheses list i like it for my 6 year old daughter as its tiny but was quoted $8000 by supplier , no way can i afford that
thanks cameron
Yes the Cellnovo is on the Prostheses List.
Australian Insulin Pumps – November 2018
I would rather prefer Omnipod for my 2 years old son with t1d. Why Australia prioritizing their financial system over someone well being?