Diabetes technology is varied, and in this instance insulin pumps. And in many ways this can end up confusing people. For example I see a lot of comments online by people saying things like
“I was told we can’t use Fiasp in pumps.”
Unfortunately that’s not true. Well, to be more precise: It Depends!
Multiple layers of “It Works”
- At a very basic level, we can use whatever actually works.
Of course we do want to be confident that it’s going to behave as we expect! - At another level, the TGA approval for each insulin says whether it’s an “approved use” in an insulin pump.
That is: it’s been tested, the evidence was submitted to the TGA, and the TGA determined that they would give the blanket “it’s OK” for that insulin to be used in pumps. - In addition, the pump manufacturer will have tested the insulin in their pump and decided to put their stamp on it as working OK.
Of course ideally we would get a tick at all three levels, and then everyone would be comfortable having been told that “It will work.” But we don’t always get that guarantee.
It can get a bit convoluted, but let’s see if I can lay it out.
Insulin approvals
The TGA has approved all of Humalog, NovoRapid, and Fiasp for use in insulin pumps in Australia.
It did not give approval for Apidra to be used in insulin pumps in (it’s labelled as for injections only). I gather this was largely due to the original clinical trials showing a higher incidence of site occlusions in the pumps being used (which are no longer current models, although that’s incidental).
So at this level we “can’t use Apidra in pumps”. And yet many people do use it quite successfully. For some people they can’t use NovoRapid or Humalog due to allergies and insulin reactions, but Apidra works fine for them. So if they’re going to use a pump they have no other choice for their insulin.
I myself used Apidra for some months back in 2018 (using a Medtronic Paradigm pump). Partly as an experiment to see if it was going to work faster than Humalog. I didn’t have any problems with it, although I was careful to change my infusion sites regularly.
Do keep in mind that in some other countries, their regulatory agencies have approved Apidra for use in pumps generally. In the US it’s approved for pumps, but only when the reservoir is replaced after 2 days.
At the same time, approvals in various countries change over time. In the US at first Fiasp did not have approval for use in pumps (only injections) but this was added later and they caught up to Australia.
Pump approvals
This is where things get more complicated. The general insulin approvals given by the TGA could be expected to apply to all pumps by default, but the pump manufacturers might identify exceptions to this. So let’s look at the pumps available on the Australian market.
Medtronic
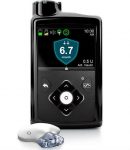 It’s relevant to point out that the reservoirs and infusion sets used with Medtronic pumps have been unchanged across many models of pump. From the Paradigm 511, 512/712, 515/715, 522/722, 554/754 (Veo), 640G, 670G, and 770G. So we would expect the same answer would apply across them all.
It’s relevant to point out that the reservoirs and infusion sets used with Medtronic pumps have been unchanged across many models of pump. From the Paradigm 511, 512/712, 515/715, 522/722, 554/754 (Veo), 640G, 670G, and 770G. So we would expect the same answer would apply across them all.
Originally they just said the pump was “intended for use with U100 insulin”.
With the 640G they specified that Medtronic had tested Humalog, NovoLog, and NovoRapid. NovoLog is of course the name used for NovoRapid in the US.
With the 670G/770G they specified:
Only use rapid acting U100 insulin (Humalog, NovoLog, and NovoRapid) that has been prescribed by your healthcare professional for use with an infusion pump.
That has Humalog and NovoRapid as examples, leaving wiggle-room for your doctor to go off-label and prescribe other insulins if required.
Accu-Chek Combo
 The Combo manual says:
The Combo manual says:
Your Accu-Chek Spirit Combo insulin pump has been developed exclusively for the continuous subcutaneous delivery of U100 short-acting insulin or fast-acting insulin analogue.
So they haven’t really restricted things at all: even covering much older insulins such as “regular” insulin (e.g. Actrapid). Almost no-one uses those older insulins in pumps today though.
Accu-Chek Solo
 The Solo manual says:
The Solo manual says:
The micropump system can be used with the following U100 insulin types: Humalog®, NovoLog®, NovoRapid®, Apidra®, Insuman® Infusat or Fiasp®.
Insuman Infusat is a more of a “regular” insulin, but we don’t get it in Australia anyway. Both Apidra and Fiasp get a mention.
YpsoPump
 The YpsoPump manual just says:
The YpsoPump manual just says:
The mylife YpsoPump® Reservoir is intended to be filled with a rapid-acting insulin at a concentration of 100 U/ml.
However the pamphlet in each box of reservoirs says:
Compatible with Fiasp®, NovoRapid®, Humalog®, Apidra®, Insulin lispro Sanofi®.
We don’t get that last one in Australia, but it’s Sanofi’s generic version of Lilly’s Humalog. Again both Apidra and Fiasp get the tick.
Tandem t:slim X2
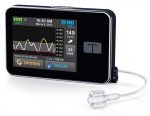 All the above pumps have a fundamentally-similar reservoir design. Basically a glorified syringe, with a plunger that’s gradually moved forwards.
All the above pumps have a fundamentally-similar reservoir design. Basically a glorified syringe, with a plunger that’s gradually moved forwards.
 The Tandem pump however is a quite different beast, with a “bladder” inside its reservoir, and a micropump which takes tiny parcels of liquid from the bladder and pushes them out the tubing. This might be a relevant point to keep in mind as we proceed.
The Tandem pump however is a quite different beast, with a “bladder” inside its reservoir, and a micropump which takes tiny parcels of liquid from the bladder and pushes them out the tubing. This might be a relevant point to keep in mind as we proceed.
The t:slim manual states:
DO NOT use any other insulin with your System other than U-100 Humalog or NovoLog/NovoRapid. Only Humalog and NovoLog/NovoRapid have been tested and found to be compatible for use in the System.
Tandem is a US company, and the t:slim came out before Fiasp had been approved for pump use in the US, so it would not be surprising if at that point they simply hadn’t put resources into testing it. And even though Fiasp had generic pump approval in Australia, the Australian agent (AMSL) could only parrot the official position of the manufacturer.
However in the time since Fiasp got US pump approval Tandem has not added any insulins to their t:slim advice.
As we saw in 2018 when the t:slim arrived in Australia, people who had been using Apidra in their pumps (including the Animas Vibe pumps many people were transitioned off with the t:slim as their only option) found out the hard way that Apidra really doesn’t work in the t:slim. Tandem obviously left it off their approval list for a reason. The liquid often seems to turn into gel and gum up the reservoir and the whole infusion set. This might be related to the pressurisation of the tiny parcels of insulin passed through the “micropump” inside the pump.
But for other insulins, we don’t know if they’ve been left off the list because Tandem simply hasn’t tested them, or if they have and found a problem. To meet their regulatory requirements they can only mention the ones they’re willing to “stand behind” and have got approved with the regulator.
Insulin-specific notes
That’s the per-pump details. It may be worth touching on a few points about specific insulins.
Humalog and NovoRapid
 These are “the standard insulins” that every pump has been tested against. They should “just work”. As long as you don’t have reactions to the insulins or to any of the excipients in the formulations.
These are “the standard insulins” that every pump has been tested against. They should “just work”. As long as you don’t have reactions to the insulins or to any of the excipients in the formulations.
Apidra
 This isn’t approved by the TGA for pump use, but I know people successfully using it in every pump type other than the Tandem t:slim X2.
This isn’t approved by the TGA for pump use, but I know people successfully using it in every pump type other than the Tandem t:slim X2.
Fiasp
 This is approved in general for pump use. The main pump that tries to exclude its use is the Tandem t:slim X2 (although as shown above some of the others don’t promote it).
This is approved in general for pump use. The main pump that tries to exclude its use is the Tandem t:slim X2 (although as shown above some of the others don’t promote it).
Keep in mind that Fiasp is essentially NovoRapid with the addition of some Vitamin B3. It’s not fundamentally different in composition.
But even in pumps that don’t specifically exclude Fiasp, it doesn’t work well for everyone. Some people report things like frequent occlusions.
But sometimes people extrapolate from that and state that Fiasp must be “crystallising” in the infusion set. Unfortunately this might not be true. As should be clear from my recent article on pump occlusion alarms, a likely scenario is that the occlusion alarms are triggered by local site inflammation inhibiting smooth infusion. It’s not certain that the infusion set or pump itself is involved in triggering the problem.
Especially when people report that it works OK for a while but then they get occlusions on the 2nd or 3rd day, it does seem likely that issues in their body are implicated more than the pump itself.
Fiasp does work well in pumps for many people (myself included). Some people find they can avoid occlusion alarms by changing the infusion site every 2 days rather than 3, but for many of us it works fine with the normal 3.
I even know some people using Fiasp in the t:slim pump. They generally do take care to “out-gas” the insulin before filling the pump. With the Combo pump I found that tiny bubbles tended to form in the Fiasp in the 8-12 hours after filling, but after flushing them out no more formed. For me those bubbles would cause gaps of no-insulin delivery (which made dosing very hit-n-miss).
I have a theory that if bubbles were making their way through the Tandem micro-pump mechanism they might trigger false occlusion alarm reports, leading to some of the issues with Fiasp.
Lyumjev
 This is the accelerated form of insulin lispro (which we usually know as Humalog). I’m mentioning it here even though it’s not available (or even approved) in Australia yet, mainly because I think (hope) it will be the next ultra-rapid insulin we might get. It’s a lot faster (both turning on and then turning off) than Fiasp, and many loopers are having success with it overseas.
This is the accelerated form of insulin lispro (which we usually know as Humalog). I’m mentioning it here even though it’s not available (or even approved) in Australia yet, mainly because I think (hope) it will be the next ultra-rapid insulin we might get. It’s a lot faster (both turning on and then turning off) than Fiasp, and many loopers are having success with it overseas.
It’s become available in the US and Europe over the last year, and is being used by many pumpers. The pump manufacturers don’t seem to have published any advice about its use yet though.
Lyumjev is essentially Humalog accelerated with the addition of a vasodilator, and there’s a lot of evidence that people using that same vasodilator in other therapy applications often have some initial site irritation (which usually subsides). So I won’t be surprised if that increases the frequency of occlusion alarms for some people. Unfortunately they might get confused and automatically blame the pump for that.
We’ll see what eventually happens!


Seems like Fiasp is good to go in medtronic 640G pumps which is great David. NR takes too long to catch up with the food intake and consequently impatience comes into play and you have more insulin and then eventually the NR overtakes the food plus some and you dive into a low. I think some of the answer with NR is to have it at least 20 mins before you eat.
They haven’t got regulatory sign-off that it’s “approved”, but I do know of many people successfully using Fiasp in those Medtronic pumps.
I use Apidra in my pump (Combo, used it before in Animas too), usually for more than 3 days: I do change set every 3 or so days, but I change reservoir only after its empty (4-5 days).