Next month we get a new insulin in Australia! Novo Nordisk’s Fiasp is finally arriving (it received TGA approval almost 2 years ago).
The Department of Health has announced that as of the 1st of June, Fiasp will be available through PBS prescriptions. It’s actually one of only a few additions to the PBS list this month. Usually there are lots of modifications (most of which don’t apply to diabetes) but at the moment the government is in “caretaker mode” during the federal election so it seems impressive that any modifications happened this month!
What is Fiasp?
Fiasp (“Faster Insulin ASPart”) is Novo’s ultra-fast-acting insulin. Effectively it’s NovoRapid (“insulin aspart”) with the addition of things including nicotinamide (a form of Vitamin B3) which accelerates the absorption.
It’s advertised as having a fast-enough onset of action that you don’t have to pre-bolus meals. Here’s one of the graphs from Novo’s EMA submission for Fiasp:
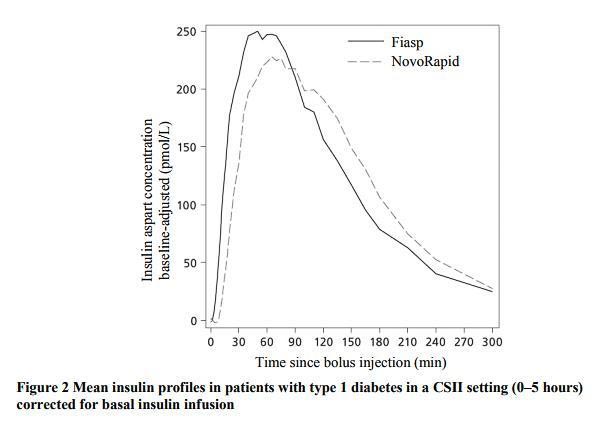
Keep in mind that this insulin is the same stuff as NovoRapid. Thus the overall activity time is the same. But because more of the insulin is “used up” early on (where the solid line is higher than the dashed line) it becomes less significant than NovoRapid once you’re past that peak.
Thus we end up with an insulin that is “faster on” as well as slightly “faster off”. Sounds great, right? Unfortunately it’s not quite that simple, as there’s more to the story.
It won’t suit everyone!
Fiasp has been available in Europe since early 2017, and the US since early 2018. As a result there’s been quite a bit of experience with it accumulated by users.
It received approval in EU markets based on some early studies which showed it to have benefits for MDI (Multiple Daily Injection) users, and “non-inferiority” for pump users. However the studies did not show significant advantages for pumpers (as discussed in this Diabettech article). And in the fine print of the study results we can see there were a number of participants who experienced problems.
In the abstract of one short-term study they just say “Unexplained hyperglycemia and premature infusion set changes were more common with FIASP than with insulin aspart.” Keep in mind that most of the early studies were only over periods of weeks rather than months.
Anecdotal evidence over the last two years shows that in pumps it works great for some people, but not at all for others. For some people their infusion sites get irritated and each stop working after 2 days (or less), or sometimes after months their insulin requirements go up dramatically and they have to eventually switch back to another insulin (luckily their insulin sensitivity usually returns quickly). The nicotinamide being constantly infused into the body seems the likely culprit.
It may be educational to read through Tim’s Fiasp articles at Diabettech.
For other people using pumps it continues to work fine, with all the speed advantages.
My own experience
The promise of an insulin that has “faster on and faster off” response seems wonderful for a closed-loop pump system, where any reduction of the latencies in the system can improve the responsiveness of the system. When Fiasp received TGA approval back in July 2017 I had already been looking at closed-loop systems for a while, and that was actually one of the prompts for me to pull my finger out and build mine so I would be ready when Fiasp became available. But in the meantime I continued to use Humalog (and the results there have been great!).
 Through some overseas travel in early 2018 I did get to use Fiasp in my closed-loop system for a while. Unfortunately my results were not great. And I did not have a lot of success in tuning the system before I ran out of supply and had to go back to Humalog. But given the mixed experiences of looping friends overseas, and concerns with the possible side-effects of long-term nicotinamide infusion, I have been very happy to have nice and reliable Humalog to depend on!
Through some overseas travel in early 2018 I did get to use Fiasp in my closed-loop system for a while. Unfortunately my results were not great. And I did not have a lot of success in tuning the system before I ran out of supply and had to go back to Humalog. But given the mixed experiences of looping friends overseas, and concerns with the possible side-effects of long-term nicotinamide infusion, I have been very happy to have nice and reliable Humalog to depend on!
David’s Fiasp summary
It seems reasonable to summarise Fiasp as:
It makes a great bolus insulin. Thus it’s great for MDI use.
But as a “basal” insulin (continuously infused through a pump) it has concerning risks.
The medical powers-that-be have approved it for use, but I’m being a bit more cautious.
I do plan to test Fiasp in my loop again soon, so don’t be surprised to see updates on this site in the coming weeks and months.
Red and yellow
Note that in early 2018 Novo changed the packaging of Fiasp from the original yellow for safety reasons. It seems adverse events had occurred when users became confused between Fiasp pens and other insulins such as long-acting Tresiba which has a very pale green highlight. In poor light and especially with red-green colour blindness, that was probably a significant safety mistake that Novo won’t make again!

Unfortunately today any images you may see of all-yellow Fiasp containers are obsolete.
Available forms
Around the world insulin is available in multiple packaging formats. But with Fiasp we won’t get all of those in Australia. At least, not yet.
10ml vials
 These are used either for injection via syringe or for filling pump reservoirs.
These are used either for injection via syringe or for filling pump reservoirs.
These will be available in Australia.
3ml pre-filled pens
Pre-filled FlexTouch pens will be available. Ryzodeg also comes in FlexTouch pens. Note that these pens are quite different to the FlexPen that NovoRapid comes in.
 The FlexTouch is “chunkier” than the FlexPen. But its major design difference is that as you dial up the dose, the plunger doesn’t extend from the pen. This means that for people who need to dial up large doses, they don’t need to stretch their thumb as far to reach the release button.
The FlexTouch is “chunkier” than the FlexPen. But its major design difference is that as you dial up the dose, the plunger doesn’t extend from the pen. This means that for people who need to dial up large doses, they don’t need to stretch their thumb as far to reach the release button.
When you do press the release button, a spring inside the pen delivers all the dose at once. Unlike the FlexPen you don’t get to control the injection speed.  Personally I don’t like this design (I had to deal with similar back in my days of using Lantus with the AutoPen 24) but pen users of Fiasp will get no choice. I referred to one of the issues (forcing a large volume of insulin quickly through ultra-fine needles) in my “What’s in a needle?” article last year.
Personally I don’t like this design (I had to deal with similar back in my days of using Lantus with the AutoPen 24) but pen users of Fiasp will get no choice. I referred to one of the issues (forcing a large volume of insulin quickly through ultra-fine needles) in my “What’s in a needle?” article last year.
3ml Penfill
 Apparently the 3ml Penfill cartridges will not be available in Australia. At least not initially.
Apparently the 3ml Penfill cartridges will not be available in Australia. At least not initially.
This means that Fiasp will not be useable in pens like the NovoPen Echo (which provides 0.5U dosing increments as well as a last-dose memory) or the NovoPen 4 (or the upcoming NovoPen 5 or the “connected” pens Novo recently demonstrated in Europe).
Pen users will have to take up a lot more room in their fridges and bags for the pre-filled FlexTouch pens, and generate a lot more waste with the disposable pens.
If you use MDI and need dosing to 0.5U increments, for now to use Fiasp you would have to go back to using syringes.
These cartridges are also useful to pump users, as they can be used to fill pump reservoirs as well as providing a convenient emergency injection backup when inserted into a pen.
Hopefully the Penfill cartridges will be added to PBS in the future. They were included in the submission to PBAC that resulted in a recommendation last November to add Fiasp to the PBS, so it’s surprising to see them omitted from the June update.
Roll on June…
The PBS will update at the start of June, so theoretically from that date doctors can write Fiasp prescriptions and pharmacies can fill them. However I do know from past experience that some medical clinics no longer use hand-written prescription pads, and there can be some slight delays getting their computer systems updated. Just be patient…


I will be new to this needle
Just been given a script for this, bit annoying about not being able to inject .5, hopefully it’s helpful for post meal spikes
I’m irritated that the 3ml Penfill isn’t available as I need ti use the pen with 0.5 increments.
This is discussed further in this article.
Cut the pen apart when you have finished and compare the vials to a penfill vial. Look very carefully. What is the difference? Think outside the box. I want .5 units too.
Jane, see my reply to the previous comment.
I am a long time insulin pen user and am very upset that fiasp is not available for penfil. The flex pens take up so much spacemin the fridge and the waste in plastic pens makes me upset as I gate having all this rubbish associated with my insulin use. All insulin should be produced in penfils these days. I hate syringes and the large 10ml vials – the 3ml pencil vials are a cost effective way for hospitals to use insulin and without the enormous waste of flex pens. Sure the pens are ok for some but I hate their size and feel. Travel is made much more difficult due to the space required to store these and keep them at the correct temperature. Penfils are the way to go and URGENTLY needed here in Australia. They are available overseas for fiasp. Bring them to Australia too and give us the freedom we deserve. It is hard enough coping with the everyday living of diabetes. Stop restricting us further by not supplying all insulin in pencil.
Hi Beth. Hopefully you’ve the other Fiasp articles on this site by now. There’s one that might describe a workaround for you.
Have just started fiasp a month ago and will be taking to my Endo to go back to novorapid (slow) I’ve have a constant headache the whole time using it and while it does work I’ve also been getting the most random highs and lows I’ve ever had. Massive spikes for no particular reason and drops that are unforeseen.
The whole time I’ve been in it I’ve felt unwell almost like a consistent high reading but the bsl say otherwise. Way too much trouble than it’s worth, I’ll deal with novoslow and work around it.
I should point out that after 41 years with t1 this is the first period of time I’ve really struggled to have control and felt unwell because of the diabetes.
That’s a shame!
You didn’t keep some Novorapid in the fridge to switch back to?
When I transitioned to Fiasp (from Humalog) I switched back and forth (>1 month each time) while I worked out its behaviour and felt comfortable I had an immediate fallback if it all fell apart.
I just started using this 6 days ago in my Medtronic 670g pump (which I am already having issues with in terms of it seemingly not delivering some meal/correction boluses!) and after a few days it was a BIG fail. I spent all day yesterday between 12-14mmol, rising constantly without eating 🙁 and then after a generous bolus for an all protein/low carb veggie meal, BOOM 20mmol and even an injection of Fiasp didn’t resolve the hyperglycaemia! Back to Novorapid for me 🙁